Good Manufacturing Practices (GMP) are the bedrock of producing safe, high-quality foods and ensuring compliance with regulatory mandates. While the specifics of GMP can often seem like an alphabet soup of acronyms and regulations, mastering GMP is essential for food manufacturers, quality control professionals, and compliance officers. It remains central to protecting public health, enhancing business agility, and maintaining a competitive edge.
This comprehensive guide will explore the multifaceted world of GMP in the food industry. Prepare to unravel the intricacies of GMP, its role in daily food manufacturing operations, and how to beef up your GMP compliance program to be not just compliant but exceptional.
What is GMP?
GMP stands for Good Manufacturing Practices, a system mandated by regulatory agencies to ensure that every aspect of the manufacturing process is controlled correctly to minimize the risk of error that could harm the consumer or spoil the product.
If you’re wondering what GMP certification is, it’s a formal recognition that a manufacturer adheres to these rigorous standards, covering sanitation, hygiene, quality control, and documentation, all crucial for ensuring food is produced under hygienic conditions.
What is the History of GMP?
The origins of Good Manufacturing Practices (GMP) can be traced back to the early 20th century, a time when food and drug safety was a growing concern amid rapid industrialization.
The catastrophic event that precipitated the formation of GMP was the 1937 Sulfanilamide disaster in the United States, where a pharmaceutical company’s lack of safety testing resulted in the death of over 100 patients, many of whom were children. This tragic event led to the enactment of the Federal Food, Drug, and Cosmetic Act (FD&C Act) in 1938, which established more stringent regulations for food and drug products.
The concept of GMP was formally introduced in the 1960s by the World Health Organization (WHO) as part of its efforts to establish international standards for drug production. The principles were later adopted and adapted by various regulatory agencies worldwide, including the U.S. Food and Drug Administration (FDA), to apply to multiple industries, including the food industry.
Since its inception, GMP has evolved significantly to address the complexities of modern food production and distribution. Early GMP regulations focused primarily on basic sanitation and manufacturing practices. However, with advancements in food technology, supply chain logistics, and an increased understanding of foodborne pathogens, GMP standards have expanded to include comprehensive risk management approaches, detailed record-keeping, and stringent quality control measures across all food production, processing, and distribution stages.
Today, GMP is a dynamic and integral component of global food safety systems, incorporating the latest scientific research and technological advancements to ensure the highest levels of consumer safety and product quality. Regulatory agencies continually update GMP guidelines to tackle emerging food safety challenges, making GMP compliance and certification an ongoing, proactive process for food manufacturers worldwide.
What is the Importance of GMP in Ensuring Quality and Safety?
GMP principles are critical for maintaining consumer trust and upholding food companies’ reputations. GMP certification fosters consumer health and satisfaction by focusing on practices that reduce contamination risks and ensure that products are safe and of consistent quality.
What are the Principles of GMP?
The principles of GMP are overarching guidelines covering every facet of production. They encompass the following critical areas:
Building and Facilities: This principle involves designing and maintaining physical structures to minimize contamination risks, support proper sanitation, and ensure a logical flow of materials and personnel to avoid cross-contamination.
Personnel: The focus here is on ensuring that all individuals involved in manufacturing, including employees and contractors, are qualified, properly trained, and understand their roles in maintaining product quality and safety.
Sanitation and Hygiene: This principle dictates the implementation of comprehensive sanitation procedures to prevent microbial, chemical, and physical contamination. It encompasses staff’s personal hygiene requirements and cleanliness protocols for the facility.
Equipment: This involves ensuring that all equipment used in the manufacturing process is appropriately designed, maintained, cleaned, and calibrated to prevent contamination and ensure consistent product quality.
Process Validation: The process validation principle requires that manufacturers scientifically prove that their manufacturing processes can consistently produce outputs that meet quality standards.
Cleaning and Maintenance: This principle emphasizes the importance of regular cleaning and maintenance schedules for facilities and equipment to prevent contamination and ensure operational efficiency.
Documentation: Comprehensive and accurate documentation is crucial for traceability, validation, and accountability. This principle requires maintaining detailed records of all aspects of the manufacturing process, from raw materials to finished products.
Quality Assurance (QA): QA is a principle that involves a series of planned and systematic activities implemented within the quality system to provide confidence that a product will satisfy quality requirements. It covers everything from material selection to final product inspection.
Validation of Processes and Procedures: Similar to process validation, this principle extends to all procedures and processes throughout the manufacturing cycle, requiring that they are followed and monitored to meet quality standards consistently.
Warnings and Recalls: This principle ensures mechanisms are in place for the rapid and effective identification and management of products that do not meet quality and safety standards, including customer notifications and product recalls if necessary.

Document Control in GMP
Document control is essential to GMP compliance. It ensures teams follow only the most current, approved procedures, minimizing errors, inconsistencies, and regulatory risks. From batch records to sanitation SOPs, every document must be up-to-date, versioned, and traceable.
Staff unknowingly followed an outdated cleaning protocol in one bakery facility, resulting in an allergen cross-contact incident. After adopting a digital GMP document control system, all updates triggered alerts and required staff acknowledgment. This simple shift eliminated similar incidents and improved audit readiness.
Strong document control isn’t just about compliance; it’s about protecting your brand and consumers with clarity, consistency, and accountability at every step.
GMP’s Role in Preventing Contamination and Errors
GMP Principles are a shield against product recalls, contaminated products, and the erosion of public trust.
The practices that underpin GMP prevent:
- Cross-contamination: The transfer of harmful bacteria or substances from one surface, food, or object to another, potentially causing foodborne illnesses.
- Incorrect Labeling: Failure to accurately describe the contents of food products, leading to misinformation and possible health risks for consumers.
- Unintended Presence of Allergens: The accidental inclusion of allergenic substances in foods poses serious risks to individuals with specific food allergies.
- Pathogen Spread: The dissemination of disease-causing microorganisms in food or environments, leading to outbreaks of foodborne illnesses.
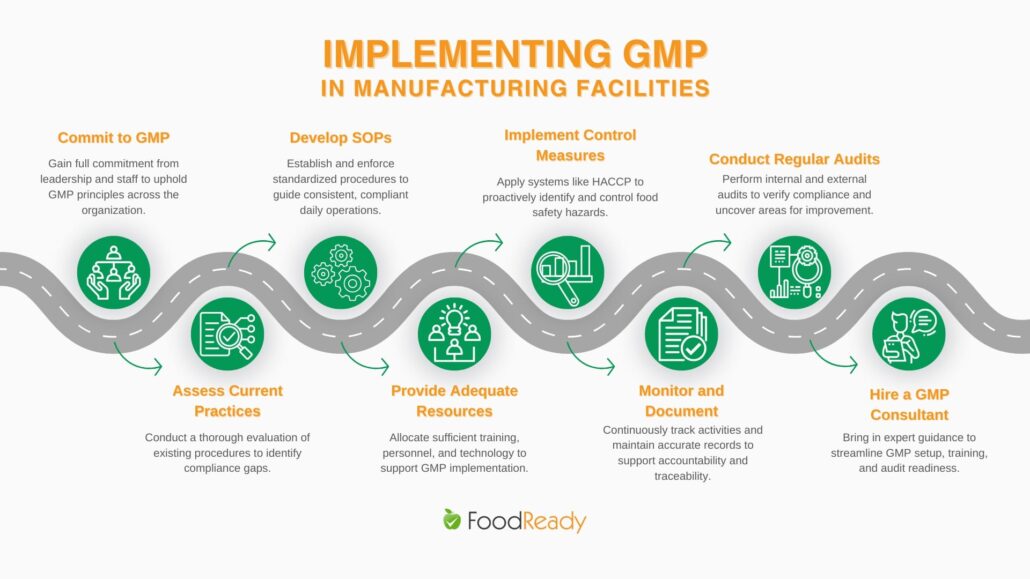
Steps to Implement GMP in Manufacturing Facilities
- Commit to GMP: Ensure all stakeholders understand and are committed to GMP principles.
- Assess Current Practices: Perform a comprehensive review of existing processes to identify gaps.
- Develop Standard Operating Procedures (SOPs): Create clear, concise SOPs and have systems in place to ensure the staff follows procedures.
- Provide Adequate Resources: Invest in the necessary technology, training, and personnel to support GMP compliance.
Implement Control Measures: Introduce measures such as HACCP (Hazard Analysis and Critical Control Points) to manage and control food safety risks. - Monitor and Document: Regularly monitor processes and maintain detailed documentation of all activities.
Conduct Regular Audits: Internal and third-party audits can provide valuable insights and help maintain compliance. GMP audit software could greatly help in this step. - Hire a GMP Consultant: Adding the expertise of a GMP Consultant will speed up the process of getting your staff and facility GMP compliant and audit-ready for GMP certification.
Strategies for Effective Quality Control
Effective quality control is a linchpin in GMP compliance. To instill such strategies, companies must:
Having a Systematic Approach: This involves developing a clear plan for managing quality throughout the entire production process. For instance, a food manufacturer might structure their approach around the PDCA (Plan-Do-Check-Act) cycle, planning quality goals, implementing strategies to achieve them, checking if the objectives are met, and then acting on any areas for improvement.
Using Testing and Analysis: Food companies often employ microbiological testing and chemical analyses to ensure products meet safety standards. An example could be using high-performance liquid chromatography (HPLC) to test for the presence of toxins or allergens in food products.
Employing Statistical Process Control (SPC): SPC involves using statistical methods to monitor and control a process. A beverage manufacturer might use control charts to monitor the pH levels of drinks over time, quickly identifying any deviations from the norm that could indicate a quality issue.
Implementing Inspection Techniques: Visual inspections, metal detectors, and X-ray systems are standard tools for identifying physical contaminants in food production. A bakery, for example, might use metal detectors to ensure that no metal shavings have contaminated their products during the mixing or packaging processes.
What is the Role of Quality Assurance in Maintaining GMP Compliance?
Quality assurance is not just about checking boxes; it is a proactive role in ensuring that GMP principles are followed.
QA in GMP Compliance includes:
Regular Review of GMP Guidelines: This activity involves periodically reviewing the GMP standards and guidelines to ensure that the practices are up-to-date. Regulations and standards can change, and ensuring compliance with the most current guidelines is crucial.
Training and Education: To maintain GMP compliance, ongoing training and education for staff at all levels within the manufacturing process are essential to keep up with changes in GMP standards, technology, and procedures.
Root Cause Analysis: When inconsistencies or deviations from quality standards are detected, conducting a root cause analysis is crucial. This detailed investigation identifies the fundamental reason for the problem, enabling the company to implement corrective actions that prevent recurrence.
Continuous Improvement Initiatives: Systematic efforts to improve processes, products, or services constitute continuous improvement initiatives. In the context of GMP compliance, these initiatives involve refining SOPs, enhancing quality control measures, or adopting new technologies to improve product safety and quality.
GMP vs HACCP vs SQF: What’s the Difference?
Navigating the sea of safety certifications and standards in the food industry can feel overwhelming. Good Manufacturing Practices (GMP), Hazard Analysis and Critical Control Points (HACCP), and Safe Quality Food (SQF) are three of the most recognized systems, but they serve different purposes within a unified goal: protecting public health through safe food production.
Understanding how these systems align and diverge is essential for building a comprehensive food safety and quality program.
Think of them as layers of protection: GMP is the foundation, HACCP is the risk-based control system, and SQF integrates both into a globally benchmarked certification.
Here’s how they compare:
| Standard | Focus Area | Scope | Mandatory | Certification | Best For |
|---|---|---|---|---|---|
GMP (Good Manufacturing Practices) | Hygiene, sanitation, operational conditions | Covers facility design, personnel, equipment, and cleanliness | Yes (regulated by FDA, USDA, etc.) | Not always required, but GMP-compliant status is often verified during audits | All food manufacturers, especially during the early compliance stages |
| HACCP (Hazard Analysis & Critical Control Points) | Identifying and controlling food safety hazards | Process-focused; evaluates biological, chemical, and physical risks | Yes (FDA FSMA requires HACCP for many sectors) | Certification available via accredited bodies | Companies producing high-risk food or exporting globally |
| SQF (Safe Quality Food) | Integrated food safety & quality management | Includes GMP and HACCP with added quality criteria | No (voluntary), but highly recognized by GFSI | Yes – third-party audited certification program | Businesses seeking global recognition, retailer approval, or competitive brand positioning |
These programs aren’t mutually exclusive for a food manufacturer aiming to grow and expand into broader markets; they’re sequential and complementary. Master GMP, implement HACCP for risk control, and use SQF to demonstrate excellence across the supply chain.
What are the Challenges in Achieving GMP Compliance?
Employee Turnover: High employee turnover rates can disrupt the consistency of GMP practices. For instance, when experienced workers leave, their replacement might take time to become familiar with the meticulous standards required, leading to potential errors or oversights in the production process.
Evolving Regulations: The constant evolution of regulatory standards can be a challenge, as companies must continually adapt their procedures to remain compliant. Recent updates in digital record-keeping requirements, for example, demand significant changes in how companies manage their documentation.
Maintaining Documentation: Ensuring that all records are up-to-date and accurately reflect current practices is critical for GMP compliance. Proper documentation includes batch records, quality control reports, and equipment maintenance logs. A lapse in this area, such as failing to record a modification in the manufacturing process, can result in violations during audits.
GMP Compliance Solutions
Effective Change Management: Adopting new processes, technologies, or operating procedures requires significant organizational adjustments and acceptance by staff.
GMP Technology Adoption: The introduction of advanced technologies is crucial for maintaining GMP compliance, especially in tracking and documenting processes.
Streamlining Document Control: GMP compliance necessitates meticulous documentation to demonstrate adherence to standards. Managing an extensive volume of documents, ensuring their accuracy, and keeping them up to date can be overwhelming, leading to potential non-compliance.
Continuous Training: The dynamic nature of GMP standards requires ongoing GMP training for employees. It’s important to inform the workforce about the latest guidelines and practices amidst their routine tasks and responsibilities.
Realignment of Culture and Values: For GMP principles to be effectively integrated into daily operations, a cultural shift within the organization is often necessary. Fostering a culture that prioritizes quality and compliance above all can be challenging, especially in well-established environments resistant to change.
Need support navigating GMP compliance?
FoodReady offers all-in-one GMP consulting and food safety software designed to simplify compliance, streamline document control, and easily train your team.
Navigating GMP Compliance in Small to Medium-sized Enterprises (SMEs)
For SMEs, GMP compliance can seem daunting. Specific strategies for success include:
- Making Incremental Changes
- Prioritizing Risks
- Leverage Collective Knowledge and Experience
- Utilize Templates and Guides
- Seamless Communication Across All Levels
The strategies outlined above can significantly enhance GMP compliance by ensuring that quality and safety are embedded into every aspect of the production process. By having a systematic approach, such as the PDCA cycle, companies can anticipate and mitigate potential quality issues before they arise.
Employing testing and analysis, along with statistical process control, allows for a data-driven understanding of the production process, ensuring that products consistently meet the established safety standards. Inspection techniques serve as a final checkpoint, catching any anomalies that may have bypassed prior controls.
Companies create a proactive culture of quality by implementing rigorous quality assurance practices, including regular review of GMP guidelines, ongoing training and education, root cause analysis, and continuous improvement initiatives. This culture does not just react to quality issues as they occur but actively works to prevent them, thereby maintaining high product safety and efficacy standards.
Furthermore, addressing common GMP compliance challenges through effective change management, technology adoption, streamlined document control, continuous training, and realignment of culture and values fortifies the foundation of GMP compliance.
By adopting these strategies, small to medium-sized enterprises (SMEs) can overcome the perceived enormity of GMP compliance, transforming it from a daunting obligation into an integral part of their operational excellence. This holistic approach improves compliance and drives efficiency, enhances product quality, and ultimately ensures customer satisfaction and trust.
The Process and Importance of GMP Audits
GMP audits are not just a regulatory requirement but an invaluable tool for improvement. They include:
- Preparing for an Audit: This initial step involves gathering necessary documents and ensuring compliance with relevant standards.
- Conducting the Audit: In this phase, auditors meticulously examine financial records, processes, and controls to assess their adequacy and effectiveness.
- Feedback and Corrective Actions: Following the audit, feedback is provided, highlighting areas of improvement and necessary corrective actions to enhance operations.
- Continuous Learning and Adaptation: Organizations should commit to ongoing learning and adaptation, leveraging audit findings to foster continuous improvement and adapt to changing environments.
Are you worried about passing your next GMP audit?
Customize the digital solution to
your exact business operations
Future Trends: AI, IoT, and Blockchain in GMP
Artificial intelligence, the Internet of Things, and blockchain technologies are poised to revolutionize GMP compliance. How can these technologies change the future of food safety?
- Predictive Maintenance: GMP management software can analyze equipment data in real time, predicting potential failures before they occur. This allows for proactive maintenance scheduling, reducing downtime, and increasing efficiency in Good Manufacturing Practices (GMP).
- Enhanced Traceability: By implementing software solutions, companies can track the journey of their products from raw materials to finished goods. This traceability ensures compliance with regulations and helps to quickly identify and isolate issues in the supply chain, maintaining product integrity.
- Real-time Monitoring: Software continuously monitors manufacturing processes and environmental conditions, flagging deviations in real time. This immediate visibility ensures that products are manufactured under consistent conditions, adhering to GMP standards and ensuring quality.
- Automated Record-keeping: Software replaces the manual task of recording data with automated systems, which not only reduces the risk of human error but also ensures a secure, tamper-proof repository of all GMP-related documents and data. Digital record-keeping makes compliance audits smoother and more reliable.
GMP compliance is an ongoing commitment to the consumer, the industry, and global safety and quality standards. GMP requires dedication, resources, and an unwavering focus on continuous improvement. By understanding, implementing, and adapting the best GMP practices, food manufacturers and their teams can meet regulatory requirements and build a legacy of trust and excellence in the food industry.
Do you want to go beyond just understanding GMP principles? Implementing a comprehensive GMP program in your food manufacturing operation is the definitive step toward securing the future of your brand. Your consumers deserve no less than your best effort in GMP compliance, and the food industry growth is intricately linked to these sound practices.
It’s time to take your GMP knowledge from theory to action, transforming your compliance program into a proactive driver of success. FoodReady food safety software and consulting services is an all-in-one solution to get your process and facility GMP compliant.
FAQs
The 5 Ps in GMP refer to People, Premises, Processes, Products, and Procedures, which are critical elements to ensure quality control and safety in the manufacturing process.
The key systems of GMP involve quality control, quality assurance, documented information (record-keeping), and sanitation and hygiene practices to ensure product safety and efficacy.
GMP (Good Manufacturing Practices) are guidelines that outline the aspects of production and testing that can impact a product’s quality. The FDA (Food and Drug Administration) is a regulatory body that enforces these guidelines in the United States.
GMP is regulated through inspections and audits by national regulatory authorities, such as the FDA in the United States, which have the authority to enforce compliance through fines, product recalls, and other legal actions.
GMP (Good Manufacturing Practices) sets the basic operational standards for manufacturing. At the same time, cGMP (Current Good Manufacturing Practices) refers to these standards as they are continually updated with the latest methods, technologies, and regulations to ensure manufacturing processes remain effective, safe, and up-to-date. Read more about the difference between GMP and cGMP in our detailed guide.







