In the food industry, cleanliness transcends the realm of basic requirement, establishing itself as a cornerstone for maintaining the highest standards of food safety and quality.
The significance of a well-maintained environment in food operations cannot be overstated, as it directly impacts public health and the integrity of the food served. An effective cleaning schedule, therefore, is not just a procedural necessity but a fundamental aspect of operational excellence.
It ensures that every facet of the food preparation and serving process is conducted in a hygienic and safe manner, safeguarding against the risk of contamination and the spread of foodborne illnesses.
The development of an effective cleaning schedule for food safety is a meticulous process that demands careful consideration of various factors. This includes the type of food operation, the volume of traffic, the nature of food handling activities, and the specific health codes and regulations pertinent to the location.
A comprehensive cleaning schedule must encompass all areas of operation, from the preparation and cooking areas to storage, dining, and waste disposal zones. Each area requires a tailored approach to cleaning, taking into account the frequency of use, the types of food handled, and the potential risks associated with each space. The goal is to create a harmonious balance between thoroughness and efficiency, ensuring that all areas are adequately cleaned without disrupting the flow of operations.
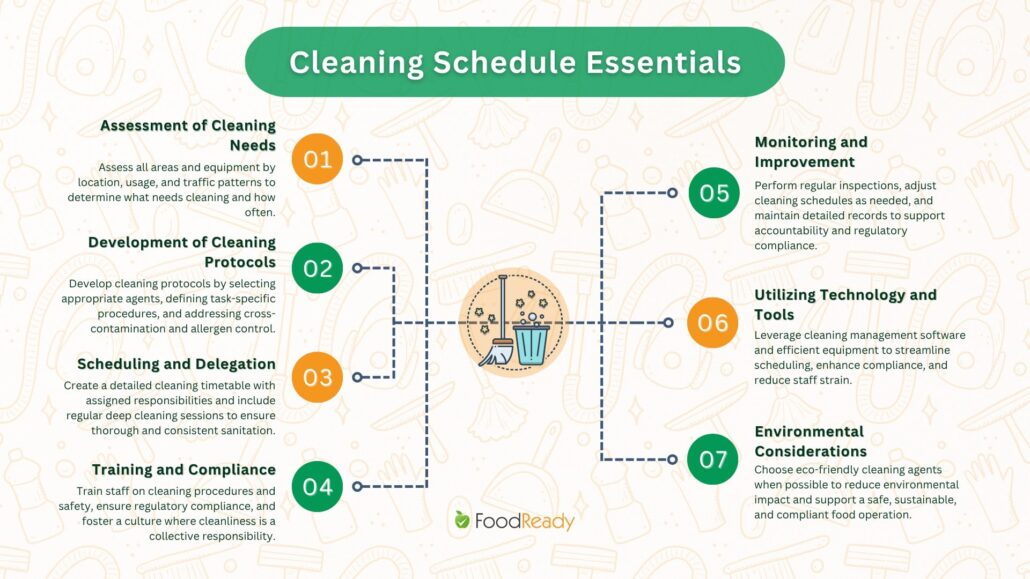
This article aims to outline the key steps and considerations involved in creating an efficient cleaning schedule tailored to food operations. This covers everything from assessing cleaning needs, establishing appropriate cleaning protocols, scheduling and assigning responsibilities, training staff, and implementing a system of monitoring and record-keeping.
The focus is not only on meeting the mandatory health and safety standards but also on fostering a culture of cleanliness and hygiene that becomes an integral part of the food operation’s identity.
Through this comprehensive approach, food businesses can ensure the highest standards of cleanliness, contributing to the health and well-being of their customers and enhancing their reputation in the industry.
The following outlines the critical steps and considerations for designing an efficient and compliant cleaning schedule in food processing environments.
1. Assessment of Cleaning Needs
Identify Key Areas: Determine all areas that require cleaning, including kitchens, dining areas, storage spaces, and restrooms.
Understand Equipment Needs: Catalogue the equipment that requires regular cleaning, like cooking appliances, cutting boards, utensils, and storage containers.
Analyze Traffic and Usage Patterns: Consider the frequency of use for different areas and equipment to determine cleaning frequency.
Zone-Based Cleaning Strategies
Cleaning strategies must go beyond general routines in food facilities, especially those handling high-risk or ready-to-eat products. Zoning involves dividing the facility into distinct areas based on contamination risk. This approach helps prevent cross-contamination between raw materials, processing zones, and finished product areas.
Zoning typically follows a risk-based model, from Zone 1 (direct food contact) to Zone 4 (external areas). Each zone requires its cleaning frequency, chemical approach, and equipment protocols. Many facilities implement color-coded tools and PPE to reinforce boundaries, ensuring that mops, brushes, and gloves from high-risk zones aren’t used elsewhere.
Here’s a simplified breakdown:
| Zone | Description | Example Areas | Cleaning Focus |
|---|---|---|---|
| Zone 1 | Direct food contact surfaces | Conveyor belts, cutting tables | Frequent cleaning and disinfection (e.g., every shift) with food-safe sanitizers |
| Zone 2 | Adjacent to food contact surfaces | Equipment frames, switches | Regular cleaning with allergen/pathogen control |
| Zone 3 | Non-food contact in processing areas | Floors, walls, and drains | Daily cleaning to control environmental hazards |
| Zone 4 | External/support areas | Offices, loading docks, breakrooms | Routine cleaning: lower risk but monitored |
A strong zoning strategy doesn’t just improve hygiene. It reinforces food safety culture by making risk visible and actionable. Staff training, signage, and tool storage protocols should all support your zoning plan to ensure consistent execution.
2. Development of Cleaning Protocols
Select Suitable Cleaning Agents: Choose cleaning agents that are effective against foodborne pathogens and safe for use in food environments.
Establish Cleaning Procedures: Create detailed procedures for each cleaning task, specifying techniques, tools, and safety measures.
Consider Cross-Contamination and Cross-Contact Risks: Implement procedures to prevent cross-contamination and Cross-Contact, especially in areas where raw and allergenic foods are handled.
Allergen Control in Cleaning Schedules
Managing allergens is a critical part of any food safety program, particularly for facilities that handle a variety of ingredients. Even trace amounts of allergenic proteins can cause severe health reactions, making cross-contact one of the most urgent risks to control. Cleaning schedules directly affect allergen prevention by ensuring that surfaces, tools, and equipment are properly sanitized between uses.
Effective allergen control starts with identifying which priority allergens are present, such as milk, eggs, soy, wheat, peanuts, tree nuts, fish, Crustacean shellfish, and sesame. Shared equipment and prep areas should follow strict cleaning protocols to remove all residues. This includes dry cleaning steps, thorough wet cleaning with appropriate agents, and detailed rinse or sanitization as required. Verification measures, such as protein swabs or allergen-specific test kits, confirm that cleaning was successful and help meet regulatory expectations.
To support these efforts, employees must be trained on allergen risks, proper cleaning procedures, and visually inspecting for residue. Integrating allergen control into your cleaning schedule doesn’t just reduce the risk of recalls, it protects vulnerable consumers and reinforces trust in your brand.
Allergen Cleaning Checklist:
- Identify and list allergens handled in the facility
- Segregate equipment when possible
- Use validated cleaning procedures for shared tools and surfaces
- Apply protein swabs or allergen test kits to verify cleanliness
- Train staff on cross-contact risks and cleaning protocols
- Log allergen-specific cleaning activities for audit purposes
3. Scheduling and Delegation
Create a Cleaning Schedule: Develop a timetable that clearly outlines when each cleaning task should be performed, whether it’s hourly, daily, weekly, or monthly.
Assign Responsibilities: Allocate cleaning tasks to specific staff members or teams to ensure accountability.
Incorporate Deep Cleaning Sessions: Schedule periodic deep cleaning sessions for thorough sanitization.
4. Training and Compliance
Train Staff: Conduct training sessions to educate staff on the importance of cleanliness, proper cleaning techniques, and safety protocols.
Ensure Compliance with Health Regulations: Make sure that cleaning procedures comply with local health and safety regulations.
Create a Culture of Cleanliness: Foster a workplace culture where cleanliness is a shared responsibility and a core value.
5. Monitoring and Improvement
Regular Inspections and Feedback: Conduct regular inspections to ensure adherence to the cleaning schedule and gather staff feedback for improvements.
Adjust the Schedule as Needed: Be prepared to revise the cleaning schedule based on changes in operations, feedback, or new health guidelines.
Record-Keeping: Maintain logs of cleaning activities for accountability and compliance with health inspections.
Validation and Verification of Cleaning Effectiveness
A cleaning schedule is only as effective as its results, and those results need more than visual confirmation. While surfaces may appear clean, harmful microorganisms or allergen residues can persist undetected. That’s why regulatory bodies and certification standards like the FDA, GFSI, and BRCGS require food operations to incorporate objective validation and verification measures into their cleaning programs.
Build Your Custom Cleaning Schedule Today
Take the guesswork out of sanitation. Use FoodReady’s guided templates to create a food safety cleaning plan tailored to your facility’s needs.
Verification methods include protein swabs or allergen-specific kits to ensure proper removal of allergenic residues. Microbial sampling before and after cleaning for facilities handling high-risk products provides quantitative assurance of sanitation effectiveness. When cleaning fails to meet expectations, corrective actions should be taken promptly, such as retraining staff, updating SOPs, or adjusting cleaning agents. Verification is not just about compliance but continuous improvement and building confidence in your cleaning protocols.
6. Utilizing Technology and Tools
Implement Cleaning Management Software: Consider using digital tools to track cleaning schedules, responsibilities, and compliance. We’ve curated a selection of top cleaning schedule software solutions for you to explore.
Use of Efficient Cleaning Equipment: Invest in efficient and ergonomic cleaning equipment to improve effectiveness and reduce staff fatigue.
7. Environmental Considerations
Eco-friendly Cleaning Solutions: Where possible, choose environmentally friendly cleaning agents to minimize ecological impact.
By following these steps and considerations, food operations can develop an effective and efficient cleaning schedule that ensures a safe, hygienic, and compliant environment, crucial for the success and sustainability of any food service operation.
Pest Control and Cleaning Interactions
Cleanliness and pest control are deeply interconnected. Even the most robust cleaning schedule can be compromised if pest activity goes unaddressed. Likewise, poor sanitation, particularly in waste or hard-to-reach areas, creates ideal conditions for infestations.
Integrating pest control considerations into your cleaning routine strengthens overall facility hygiene and aligns with Integrated Pest Management (IPM) strategies.
Cleaning That Supports IPM
Routine cleaning removes food sources and nesting materials that attract pests. Focused efforts should target under-equipment spaces, drains, wall-floor junctions, and storage corners; areas where crumbs, moisture, and debris accumulate unnoticed. Consistent sanitation disrupts pest lifecycles and reduces dependence on chemical controls.
Waste Handling Protocols
Waste areas require daily cleaning and sanitization, especially in hot or humid conditions. Bins should have tight-fitting lids, be emptied regularly, and be positioned away from food prep zones. To prevent harborage points and rodent access, exterior waste areas should be swept and washed frequently.
What to Watch For
Cleaning staff play a key role in early pest detection. Train them to recognize warning signs like droppings, gnawed packaging, grease trails, insect carcasses, or nesting debris. Prompt reporting of these indicators allows for swift intervention and minimizes contamination risk.
FDA’s 2025 Sanitation Guidance and What It Means for Your Cleaning Program
In January 2025, the FDA published a significant new draft guidance titled “Establishing Sanitation Programs for Low-Moisture Ready-to-Eat Human Foods and Taking Corrective Actions Following a Pathogen Contamination Event.” This guidance is relevant to any manufacturer or processor of low-moisture, ready-to-eat (LMRTE) foods, a broad category that includes peanut butter and nut butters, powdered spices, powdered infant formula, chocolate, processed tree nuts, granola bars, dry cereals, milk powders, and snack foods such as chips and crackers.
The guidance provides the FDA’s current thinking on how facilities can establish routine sanitation programs that prevent pathogen contamination events and outlines corrective action steps when prevention fails. Several of its recommendations have direct implications for how cleaning schedules should be structured.
First, the guidance draws a clear distinction between cleaning and sanitizing as two separate, sequential steps in dry processing environments. Cleaning removes residue and organic matter from food contact surfaces, while sanitizing destroys pathogens. The FDA explicitly cautions that material flush techniques, which involve pushing product or hot oil through a food contact surface to remove residue, are not adequate substitutes for proper cleaning and sanitizing. Any cleaning schedule for LMRTE facilities must reflect this distinction.
Second, the guidance emphasises the importance of controlling water in dry production environments, noting that water can enable environmental pathogen growth and facilitate the spread of Salmonella and Cronobacter, both of which have histories of establishing persistent resident strains in dry food manufacturing plants. Wet cleaning procedures, when used, should be conducted in designated areas and with controlled techniques to minimise moisture spread across the facility.
Third, the guidance highlights the limitations of relying solely on product testing as verification that contamination has been eliminated. An effective cleaning program must include environmental monitoring, documented corrective actions, and root cause investigation protocols, which should all be reflected in the facility’s master sanitation schedule.
For food manufacturers producing LMRTE products, reviewing and updating your cleaning schedule in line with this draft guidance is a proactive step toward regulatory readiness. FoodReady’s food safety software supports this process by helping teams build and manage master sanitation schedules, track environmental monitoring results, log corrective actions, and maintain the documentation records required for compliance audits.
Conclusion
In summary, creating an efficient cleaning schedule for food operations involves a multi-step approach that starts with assessing cleaning needs across various areas and equipment, considering usage patterns and potential contamination risks.
It then moves on to developing specific cleaning protocols, selecting suitable cleaning agents, and establishing detailed procedures to avoid cross-contamination. The schedule must be clearly defined, assigning responsibilities to staff members and including regular deep cleaning sessions.
Training staff in proper cleaning techniques and compliance with health regulations is crucial, as is fostering a cleanliness-focused workplace culture. Regular monitoring and adjustments to the schedule ensure continued effectiveness, while record-keeping aids in compliance and improvement.
Additionally, the use of technology for schedule management, investment in efficient cleaning tools, and consideration for eco-friendly solutions and allergen control are important aspects of the process. This comprehensive approach ensures a hygienic, safe, and compliant environment in food operations.
FAQs
Regular food safety audits evaluate adherence to predefined cleaning protocols and effectiveness. Audit outcomes often prompt adjustments to cleaning schedules, ensuring they meet industry standards and address any newly identified risks.
Digital tools offer advantages over traditional logs by providing real-time updates, reminders, and easy compliance tracking. They enhance transparency and accountability in maintaining cleaning standards but require proper integration into existing systems.
Cleaning schedules should be dynamic, increasing frequency during peak production periods when the risk of contamination is higher. Seasonal adjustments ensure that cleanliness standards are maintained despite fluctuating production volumes and different food types.
Using eco-friendly cleaning agents helps reduce environmental impact without compromising hygiene standards. These agents are significant in food production to avoid chemical contamination, align with consumer preferences for sustainability, and comply with environmental regulations.
In January 2025, the FDA released draft guidance specifically addressing sanitation programs for low-moisture, ready-to-eat (LMRTE) foods, covering products such as peanut butter, powdered spices, processed tree nuts, dry cereals, and chocolate. The guidance reinforces several practices that should be reflected in cleaning schedules for these facilities. It clarifies that cleaning and sanitizing must be performed as two separate, sequential steps and that material flush techniques are not acceptable as a primary sanitation method. It also stresses the importance of controlling water use during wet cleaning in dry production environments, where excess moisture can enable the growth and spread of environmental pathogens such as Salmonella and Cronobacter. Facilities producing LMRTE foods should review their current master sanitation schedules against the draft guidance to ensure their cleaning frequencies, chemical protocols, and verification procedures are aligned with the FDA’s current recommendations.







